Surgery Vs Conservative Management of Femoral Acetabular Impingement Syndrome (FAIS)
Reflections on the 2018 “FASHION” Trial
In Australia, between 2010 and 2013, the number of people undergoing hip arthroscopy increased by over 50% (Medicare Australia, Medicare Benefits Schedule Item Statistics 2014: http://medicarestatistics.humanservices. gov.au), and in the United States it increased 6-fold between 2006 and 2010. [1] However, high-level evidence of the efficacy of both surgical and nonsurgical interventions for patients with FAIS and/or labral tears is lacking.
Rigorous research designs involving placebo controls, randomization, allocation concealment, and double blinding are considered gold standards for medical interventions, but unfortunately are less common within surgical interventions than in non-invasive interventions. [2] Surgical procedures have many factors that could contribute to placebo responses. [3, 4] In a recent systematic review, large effects of placebo surgery and other invasive procedures, particularly in the field of pain related conditions, were observed. [2]
Many patients undergoing hip arthroscopy have not been informed of the absence of evidence supporting a beneficial effect of surgical procedures compared to other treatment options for FAIS.
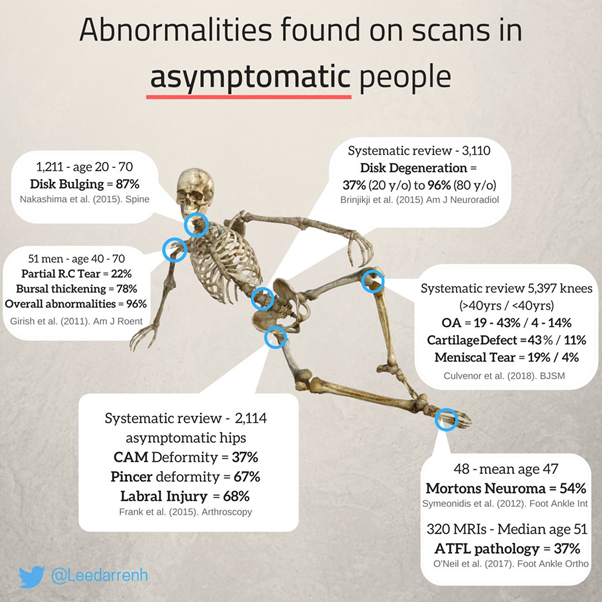
While evidence is lacking to support the use of surgical intervention, its biological plausibility is also under question, given that completely asymptomatic individuals often have morphology typically seen in FAIS (see image below), and there is a lack of association between morphological changes of the hip, symptoms, and dysfunctions. [5, 6] And despite this, the number of hip arthroscopies has risen sharply. [7]
So why are so many hip arthroscopies being performed?
As Morgan [2018] describes, in the field of medicine, there has historically been the requirement for significant evidence to disprove something (i.e. remove an ineffective treatment from the market); but merely analogy, expert opinion or physiological narrative to adopt something. [8]
This is precisely what is wrong in the field of expensive and invasive treatments or chronic pain. Low standards for adoption, despite ample history showing the folly of this approach.
The role of industry:
Device manufacturing companies have important roles in shaping the opinions of physicians and patients. The mandate of device manufactures is to sell as much product as possible for the highest possible price. [9] For publicly traded companies, the Chief Executive Officers’ incomes are largely dictated by share prices, which are affected by perceived future profit streams and not by the magnitude of the benefits of their products to patients. [9] The influence of device manufacturing companies permeates medical education, physician thought leaders, the clinical research agenda, and practice guidelines — especially when guidelines are developed by professional societies that receive substantial funding from industry. [9]
The device manufacturing industry has the technical expertise and financial resources to dominate the clinical research agenda and design the majority of clinical trials, including the choice of outcomes that sound important but are often not (such as the use of surrogate endpoints) [10]. Industry ‘mega-trials’ have the power to detect very small gains. Selective reporting of trial results and ‘spin’ of marginal effects overstate the benefit of small gains. [11]
For example, last year a poorly designed surgical trial, with no placebo control group, and a strawman conservative group, was heralded as a success.
“The FASHION Trial”
The Positive Spin:
The lead author kicked off the social media PR, followed by enthusiastic proponents of the procedure
The positive spin put on this trial was interesting.
As it turns out, only 1 of 10 outcomes favoured arthroscopy, even with a biased trial design.
All serious adverse events related to treatment were in the arthroscopy group, including post-surgical infection.
Introduction of bias:
One of the inclusion criteria for the FASHION trial was that you were deemed appropriate for surgery by the surgeon. In other words, participants were told by the surgeon “you would benefit from surgery” but subsequently randomly allocated to physiotherapy led conservative management.
Patient’s expectation of improvement would have been significantly reduced if patients were told;
- They had a structural abnormality that is causing their pain
- They would benefit from surgery to correct the abnormality
- But were instead allocated to “exercises”
As we can see from the data, just because a person with pain has FAI morphology on imaging does not mean the imaging finding is the reason for the pain. But if the patient believes that is the reason, psychologically, the idea that non-surgical treatment will “fix” their "problem" may seem illogical. Words, images, as well as beliefs and the overall narrative of the pain experience are significant. This is a salient bias that this trial completely ignored.
Crafty cropping of graphs:
In addition to the bias allowed to freely enter, the difference in outcome was also visually amplified by cropping the graph to make the one of the left appear as though the effect size was larger. [16]
Strawman control group:
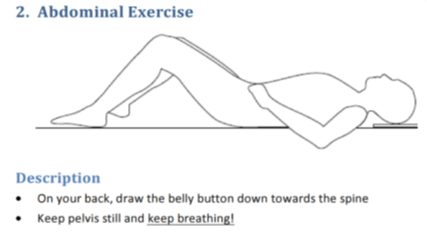
The non-surgical intervention was described as “Best Conservative Care”.
However, the exercises given primarily target hip and trunk ‘stability’ with body weight or resistance bands. The full exercise database can be found here. I have shown below some examples of what was involved.
This is precisely the sort of reductionist, biomedical “stability” focused messaging that we are trying to move away from as a profession. Telling someone that they have something structurally wrong with them based on scans, and then prescribing “stability” exercises, while ignoring psychosocial factors, is not evidence based treatment. Chronic pain is a biopsychosocial condition. The authors claim, in the title of their paper, that the conservative group received “Best Conservative Care”. This is factually inaccurate.
This article by Cheryl Lee on the Pain-Ed website explains precisely why the exercises given in the FASHON trial were not only ineffective, but may potentially contribute to chronicity in susceptible individuals:
“Hold up your fist in front of you and clench it hard.
Keeping it clenched, how far you can roll the wrist forwards and backwards? How does that feel? Comfortable? Easy? Of course not. Would you be comfortable walking around all day with the muscles of the hand, wrist and forearm held in co-contraction? Of course not.
I did the prescribed exercises, even though they made things feel tighter. I got worse and was doing less, never feeling any stronger or more stable. Physiotherapy could do nothing more for me. The chronic pain clinic told me to start using a wheelchair. It was a hell that lasted 6 years, but that is another story.
I was not broken. My core was not weak. My spine was robust and strong, not unstable. I was scared to bend, squat, or move quickly, so avoided it. My breath was shallow and ragged. I was scared about the future. This ongoing fearful situation had ramped up my body’s defences and jacked up the tension in my body, splinting me more than the exercises I had been doing.
The advice I had been given, the words that had been said, the beliefs I had taken on through my own learning had turned out to be nocebos — noxious messages with a detrimental effect on my health and well-being — that kept me locked in a prison partly of my own making.
The constant co-contractions, the fear, the movement avoidance — these had been huge factors in the pain symptoms. I learned to let go of my belly and back, and create space in my body; move with fluidity and ease; breathe fully into a soft, yielding belly; slouch and relax at the same time. I learned that I could contract my muscles with control, but also release them FULLY; that I could run, jump; ride a bike with a rounded back. I learned my spine and body were strong, and that I could get better.
I’m not an isolated case. This is universal. Let’s stop peddling old-fashioned ideas and bullshit about health. Stop selling movement and exercise by telling people they are weak and unstable. Instead, let’s empower, educate and help people to make sense of their pain, shake the labels, improve their function and move fluidly and fearlessly.” [12]
The conservative arm was setup to fail (a strawman), not only because they didn’t receive an elaborate theatrical placebo with a convincing narrative (like the surgical group), but because the treatment itself is not reflective of best care, and potentially iatrogenic.
Financial conflicts and industry influence:
As outlined in the trials declaration of interests, the lead author is personally funded by device manufactures:
Stryker is one of the top 5 leading device manufactures in the world, with a net annual revenue of $1.02 Billion. [13]
Multiple studies have shown that doctors who are personally funded by private industry tend to have more favourable views of products, and their subsequent practice behaviour is affected. [9]
By purposely designing a trial like FASHION, without a sham control group, the effectiveness of surgery is overestimated, and question marks over causality still remain.
The lead author, sponsored by one of the world’s largest device manufactures, personally;
1. Designed the trial (knowingly introducing bias that could have been eliminated with sham control)
2. Wrote the grant application
3. Drafted the final report
4. Was the guarantor for the study
5. "Decided to submit the manuscript"
6. Was personally funded (outside his submitted work) by a group set to financially benefit from a positive result from the trial.
In light of the evidence, these facts above should allow clinicians to view this study in context.
Ideally, it should be academics, free from financial conflicts of interest with industry, who design trials, apply for grants and have the final decision regarding submission of the manuscript.
Low levels of scientific rigor required for surgical interventions:
If arthroscopy were a pharmaceutical drug, the procedure would be required to undergo scrutiny of testing its safety and feasibility in phase 1 and 2 blinded placebo trials. This is obviously not the case in the field of surgery.
In other words, if hip arthroscopy for FAI was a pharmaceutical, the current standard of evidence would not permit it to market.
Additionally, once a practice has achieved market share, it is very difficult to get physicians to halt or abandon the practice, even if it is later shown to be ineffective in unbiased clinical trials. Some studies estimate that it takes up to 17 years for “medical reversal” (abandoning or scaling back on a practice after evidence says it's inappropriate). [14]
The “last resort” argument:
In spite of this clear lack of reliable evidence, this is the sort of argument I hear quite frequently from proponents of the surgery:
“Some patients with hip shape of FAI are asymptomatic and certainly don’t require treatment. Some patients with FAI improve with physical therapy, and don’t need surgery. But the group that remains in pain can benefit from properly done hip arthroscopy.”
There is no logic to this. Failed conservative management is not an adequate justification for surgery that has no evidence.
In the cited example, if we wish to engage in evidence based practice, "group that remains" needs to be enrolled in a sham RCT to determine if any benefit from "properly done" hip arthroscopy actually relates to structurally altering the hip.
Its asymptomatic prevalence alone should challenge the notion that structure equates to pain. All prior negative sham RCTs for chronic pain should also question the biological plausibility of the rationale.
The history of medical science demonstrates that invasive interventions based on bioplausibility for chronic pain are more likely to fail, rather than succeed, when subjected to blinded placebo controlled randomised trials [15].
Now we have to wonder, how did this clinical trial get published in the topmost journal?
Part II
Actual “Best Practice Conservative Care”
The biopsychosocial model
The biomedical model is defined as a model of illness that ignores psychological and social variables, and focuses only on biological pathology. This model has been a catastrophic failure for chronic pain suffers, as outlined below.
The biomedical model fails to capture the true reasons for level of disability in chronic pain suffers, because pain intensity and magnitude of disability are explained better by psychosocial factors than by pathophysiology in patients with musculoskeletal disease [1], and injury [2], particularly in a workers compensation setting [3]. The biopsychosocial model also reveals why the reductionist passive interventions offered early in chronic pain suffers treatment are usually ineffective, and often serve to actually perpetuate the recovery.
For example, experimental studies have shown that negative emotions like depression, anger, and anxiety can increase both pain intensity and unpleasantness [4]. These experimental studies are corroborated with epidemiological data suggesting that musculoskeletal trauma results in self-reported disability and pain [5], which is worsened by such psychosocial factors [6].
In addition to depression [6], both self-efficacy about pain [7] and catastrophic thinking [8] are regarded as significant psychosocial factors that can influence disability and pain intensity, and a biopsychosocial framework allows appreciation of these influences.
The biopsychosocial model also allows us to appreciate how pain experience itself changes across individuals circumstances. As pain perception is modulated by a variety of social, psychological and demographic factors [9], the biopsychosocial model enables holistic assessment and treatment algorithms, which are crucial for successful management of chronic pain [10].
Interestingly, there is compelling evidence of racial and ethnic differences in pain perception [9], low socioeconomic status is also a risk factor for chronic pain [11], and different meaning is attributed to pain in different cultures [12]. It is clear from these insights that psychological and social factors are instrumental, and a biopsychosocial approach is really the only evidence based way of navigating this terrain.
Furthermore, there is a relationship between traumatic events in childhood and chronic pain [13, 14]. A biopsychosocial approach enables early interdisciplinary management. Mitigation of psychological comorbidities is associated with improved outcomes in chronic pain [15], and a biomedical model approach would fail to consider this variable.
Additionally, injuries often occur in situations where patients assess the situation as “unjust”. Using the biopsychosocial model one can assess whether a client conceptualises the experience as unjust and see themselves as a victim [16]. A sense of ‘perceived injustice’ has been associated with fear of movement, poor rehabilitation and higher pain severity [17], higher self-reported disability [18], pain catastrophizing [19], as well as prolonged absence from work [20] in patients with chronic pain.
Additionally, patients being treated for chronic pain under workers compensation are generally more psychologically distressed, more severely disabled, are less active and have poorer outcomes than non-compensated patients[21]. As noted by Mendelson et al. [22], “If you have to prove that you are ill, not only can you not get well but you are also likely to sink further into the abyss of vulnerability and to learn ‘illness behaviour’ in the process.” An analysis of patients using a biopsychosocial approach enables the necessary multifaceted and multifactorial approach to treatment, subsequently reducing the risk of further iatrogenic insult and ineffective management.
Part IIa
Pain correlates more closely with psychosocial variables than pathology on scans
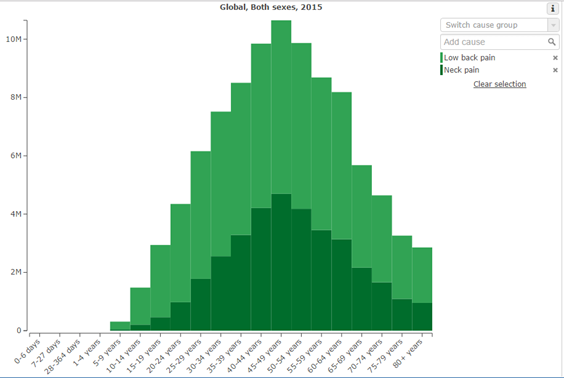
The graphs below indicate 3 things:
1. On average, musculoskeletal pain peaks in midlife
2. On average, stress peaks in midlife, and wellbeing troughs in midlife
3. Objective degenerative change on scans steadily increases throughout life, and is not reliably correlated with pain.
Therefore, there appears to be a relationship between increasing psychosocial stressors in midlife, and increased prevalence of musculoskeletal pain. This epidemiologic data correlation is reinforced by studies demonstrating that, indeed, pain intensity and magnitude of disability are better correlated with psychosocial factors than with pathophysiology in patients with musculoskeletal disease and injury [2, 3].
Following peak YLD in the 50-54 age bracket, there is a gradual tapering off to 11, 490 YLD in the 60-64 year age bracket. Interestingly, this is followed by a significant decrease over the age of 65 (post retirement age) to 8, 839, falling further over the age of 70 to 7, 123YLD, and paradoxically, falling to the lowest rates since the 15-19 year age bracket at 75-79 years.
Prevalence then spikes again over the age of 80. This spike over 80 could also be explained by the influence of both potentially reduced mobility, and potential psychosocial effects. Given the prevalence of abnormal low back pain pathology on imaging in the 80+ age group (figure 2) [4], we can rationally infer than pathology on imaging is unable to explain pain with any degree of reliability. Both psychosocial stressors and reduced movement are more likely to explain this spike. For example, stress in the over 80 age group on average increases due to negative health expectations, the threatening of personal purpose and reduced autonomy [5]. Anxiety experienced in daily life caused by fear of death in the elderly is also a potential psychosocial stressor that has been examined [6].
In all stages of life these factors and mediated by locus of control, social support, socioeconomic status and other variables [6], all which fall under the purview of the biopsychosocial construct.
Part IIb
The need for biopsychosocial interdisciplinary treatment
Evidence demonstrates that some GP’s make more than five times as many referrals per patient as others do [1], and this variability is based on provider training, years of experience, and experience with the presenting condition [2].
There is compelling evidence to suggest that a referral to specialists, including orthopaedic surgeons, early in pain management is unnecessary and may be iatrogenic [3]. Evidence for chronic pain clearly indicates that staying active, improving one’s understanding of pain, and identifying risk factors for persistent pain results in improved outcomes for most people [4]. However, given that results for invasive treatments are, on average, no better than conservative interventions [5], plus individuals with high score on psychometric testing typically do not benefit from surgery at all [6], and (dubiously) rates of surgery vary geographically according to the surgeon’s enthusiasm for surgery and the availability of MRI [7], referrals for chronic pain patients to orthopaedic surgeons (in the absence of red flags) is fundamentally put into question.
Notwithstanding, there is also evidence to suggest that the language involved in simply referring someone to an “orthopaedic surgeon” early in management can inflame the perceived threat and sense of danger around their hip (for a vulnerable person) [8]; this is particularly relevant for patients with psychosocial stressors.
Additionally, reductionist passive treatments given in isolation for chonic pain are non-evidence based treatments that completely ignore psychosocial variables, and can serve to make patients more distressed, feel more like a patient and reinforce the passivity of their role [9]. This style of treatment is also demonstrably paternalistic and disempowering; and, there is also strong evidence that passive treatments can actually harm patients [10]. Outlining to patients that passive, instead of active, management strategies are advisable can exacerbate patients fear and anxiety about being physically active, contributing to increased anticipatory anxiety about pain and (potentially) further prolonging recovery [8].
In the case of chronic pain, often many months after the initial injury (a period of time where any actual tissue damage would have long since been healed [11]), CT scans and MRI’s are ordered by treating practitioners. In the absence of suspected red flags, these scans expose patients to risk of unnecessary surgery, as any incidental hip pathology (CAM found in 37% of asymptomatic patients) can be falsely attributed to their pain [12]. Also, given what we know about verbally induced hyperalgesia and allydinya, CT or MRI results containing the words “narrowing”, and “impingement”” can worsen a patients symptoms and further circumscribe their function through fear of movement [13]. CT’s and MRI’s are also expensive, and CT’s can expose patients to unnecessary radiation [14].
How might the early stages of a patient’s management be improved?
The early musculoskeletal pain management can be improved if the patients primary health care provider can refer to one allied health practitioner (physiotherapist etc) who can have oversee the management through an evidence based, biopsychosocial, interdisciplinary framework.
Some research supports the efficacy multidisciplinary pain treatment [15]. However, given that research also indicates that early aggressive psychological interventions in the acute phase of injury may actually undermine recovery [16, 17, 18], a well-informed physiotherapist can implement an interdisciplinary approach (such as Stress Inoculation Training), thereby assisting coping and problem-solving strategies to reduce stress-related anxiety [16]. Additionally, a physiotherapist can packaged treatment to provide essential information to the client about the impact of stress on their physical and psychological wellbeing; thus facilitating improved insight, which may subsequently reduce the risk of chronicity [19].
This, combined with a ‘classification-based cognitive functional therapy’ (CFT) approach, (which may include a graduated, mindful movement based approach to exercise that emphasises decreasing fear about movement, increasing self-efficacy, self-understanding and self-empowerment, explaining pain physiology, empathetic patient-centred communication, demystifying beliefs about pain (beliefs which come from interactions with previous health care practitioners), and thus increasing the patients’ health/scientific literacy) is well supported by evidence [20], and may have prevented many chronic pain patients unfortunate slide into chronicity.
For patients with pre-existing chronicity and high degrees of psychological distress, psychological treatments based on the principle of cognitive behavioural therapy are supported by evidence [21], and mitigation of psychological comorbidities is associated with improved outcomes in chronic pain [22].
Yoga and mindfulness within a CFT framework
Honest discussion with patients about the factors outlined above is imperative in sound management FAI.
Mindfulness and yoga therapy can be applied as the graded exercise component within at CFT framework, with its techniques to modulate anxiety. Since pain appears to be amplified by anxiety through the activation of cholecystokininergic systems, strategies to reduce anxiety, such as yoga therapy, can be applied whenever pain has an important anxiety component. According to Streeter et al (2007) [23], a regular practice of yoga releases a neurotransmitter chemical called GABA. Individuals who exhibit increased anxiety have reduced levels of GABA. The authors believe that these sustained levels of GABA over a period of regular yoga practice may help the brain to rewire itself, reducing anxiety and thereby modulating the pain response. Additionally, Mindfulness Meditation is associated with lower amygdala activation, which is related to perceived threat. Pain is often the brains response perception of threat [24].
Furthermore, chronic pain is known to cause brain anatomy changes and impairments, but yoga can be an important tool for preventing or even reversing the effects of chronic pain on the brain. [25]
One of the problems in the implementation of these techniques is that most physiotherapists don’t even meet the low bar set for recommended physical activity levels that they themselves promote to patients. [26] It would be ideal if physios were able to lead their patients by example. It would likely make them better movement facilitators for their patients.
References:
Part I REFERENCES:
1. Bozic KJ, Chan V, Valone FH, 3rd, Feeley BT, VailTP. Trends in hip arthroscopy utilization in theUnited States. J Arthroplasty. 2013;28:140-143.https://doi.org/10.1016/j.arth.2013.02.039
2. Jonas WB, Crawford C, Colloca L, et al. To what extent are surgery and invasive procedures effective beyond a placebo response? A systematic review with meta-analysis of randomised, sham controlled trials. BMJ Open. 2015;5:e009655. https://doi.org/10.1136/ bmjopen-2015-009655
3. Johnson AG. Surgery as a placebo. Lancet. 1994;344:1140-1142. https://doi.org/10.1016/ S0140-6736(94)90637-8
4. Sihvonen R, Paavola M, Malmivaara A, et al. Arthroscopic partial meniscectomy versus sham surgery for a degenerative meniscal tear.N Engl J Med. 2013;369:2515-2524. https://doi. org/10.1056/NEJMoa1305189
5. Brunner R, Maffiuletti NA, Casartelli NC, et al. Prevalence and functional consequences of femoroacetabular impingement in young male ice hockey players. Am J Sports Med. 2016;44:46-53.https://doi.org/10.1177/0363546515607000
6. Philippon MJ, Ho CP, Briggs KK, Stull J, LaPrade RF. Prevalence of increased alpha angles as a measure of cam-type femoroacetabular impingement in youth ice hockey players. Am J Sports Med. 2013;41:1357-1362. https://doi. org/10.1177/0363546513483448
7. Clohisy JC, Baca G, Beaulé PE, et al. Descriptive epidemiology of femoroacetabular impingement: a North American cohort of patients undergoing surgery. Am J Sports Med. 2013;41:1348-1356. https://doi.org/10.1177/0363546513488861
8. Morgan, D. (2018, May 31). Like most of (US) medicine, we require evidence to disprove something but analogy, expert opinion or physiological narrative to adopt something. Hospital contact precautions anyone? @CDCDirector https://t.co/KFUg5NUBzN. Retrieved from https://twitter.com/dr_dmorgan/status/1002333546047041537
9. Booth, C. M., & Detsky, A. S. (2018, September 24). Why patients receive treatments that are minimally effective? Retrieved from https://www.nature.com/articles/s41571-018-0101-4
10. Booth, C. M. & Eisenhauer, E. A. Progression-free survival: meaningful or simply measurable? J. Clin. Oncol. 30, 1030–1033 (2012).
11. Boutron, I. et al. Reporting and interpretation of randomized controlled trials with statistically nonsignificant results for primary outcomes. JAMA 303, 2058–2064 (2010).
12. Lee, C. (n.d.). Home. Retrieved from http://www.pain-ed.com/blog/2018/06/18/bullshit-about-posture-causes-pain-suffering/
13. Stryker Corporation. (2018, December 03). Retrieved from https://en.m.wikipedia.org/wiki/Stryker_Corporation
14. Prasad, V. K., & Cifu, A. S. (2015). Ending medical reversal: improving outcomes, saving lives. JHU Press.
15. Jonas, W. B., Crawford, C., Colloca, L., Kriston, L., Linde, K., Moseley, B., & Meissner, K. (n.d.). Are Invasive Procedures Effective for Chronic Pain? A Systematic Review. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/30204920/
16. Jevne, J. (2018, October 24). Jørgen Jevne (@jevnehelse). Retrieved from https://twitter.com/jevnehelse
Part II REFERENCES:
1. Ring, D., Kadzielski, J., Malhotra, L., Lee, S. G. P., & Jupiter, J. B. (2005). Psychological factors associated with idiopathic arm pain. J Bone Joint Surg Am, 87(2), 374-380.
2. Crichlow, R. J., Andres, P. L., Morrison, S. M., Haley, S. M., & Vrahas, M. S. (2006). Depression in orthopaedic trauma patients. J Bone Joint Surg Am,88(9), 1927-1933.
3. Gabbe, B. J., Cameron, P. A., Williamson, O. D., Edwards, E. R., Graves, S. E., & Richardson, M. D. (2007). The relationship between compensable status and long-term patient outcomes following orthopaedic trauma. Medical Journal of Australia, 187(1), 14.
4. Sullivan, M. J. (2006). Psychological Methods of Pain Control: Basic Science and Clinical Perspectives. The Journal of Nervous and Mental Disease, 194(5), 388-389.
5. Lindenhovius, A. L., Buijze, G. A., Kloen, P., & Ring, D. C. (2008). Correspondence between perceived disability and objective physical impairment after elbow trauma. J Bone Joint Surg Am, 90(10), 2090-2097
6. Vranceanu, A. M., Barsky, A., & Ring, D. (2009). Psychosocial aspects of disabling musculoskeletal pain. J Bone Joint Surg Am, 91(8), 2014-2018.
7. Nunez, F., Vranceanu, A. M., & Ring, D. (2010). Determinants of pain in patients with carpal tunnel syndrome. Clinical Orthopaedics and Related Research, 468(12), 3328-3332.
8. Teunis, T., Bot, A. G., Thornton, E. R., & Ring, D. (2015). Catastrophic thinking is associated with finger stiffness after distal radius fracture surgery.Journal of orthopaedic trauma, 29(10), e414-e420.
9. Riley, J. L., Wade, J. B., Myers, C. D., Sheffield, D., Papas, R. K., & Price, D. D. (2002). Racial/ethnic differences in the experience of chronic pain.Pain, 100(3), 291-298.
10. Nielson, W. R., & Weir, R. (2001). Biopsychosocial approaches to the treatment of chronic pain. The Clinical journal of pain, 17(4), S114-S127.
11. Saastamoinen, P., Leino-Arjas, P., Laaksonen, M., & Lahelma, E. (2005). Socio-economic differences in the prevalence of acute, chronic and disabling chronic pain among ageing employees. Pain, 114(3), 364-371.
12. Riley III, J. L., & Wade, J. B. (2004). Psychological and demographic factors that modulate the different stages and dimensions of pain. Psychological methods of pain control: basic science and clinical perspectives. Seattle, WA: IASP, 19-41.
13. Goldberg, R. T., PACHASOE, W. N., & Keith, D. (1999). Relationship between traumatic events in childhood and chronic pain. Disability and rehabilitation, 21(1), 23-30.
14. Walker, E., Katon, W., Harrop-Griffiths, J., Holm, L., Russo, J., & Hickok, L. R. (1988). Relationship of chronic pelvic pain to psychiatric diagnoses and childhood sexual abuse. Am J Psychiatry, 145(1), 75-80.
15. Burns, J. W., Kubilus, A., Bruehl, S., Harden, R. N., & Lofland, K. (2003). Do changes in cognitive factors influence outcome following multidisciplinary treatment for chronic pain? A cross-lagged panel analysis. Journal of consulting and clinical psychology, 71(1), 81.
16. Trost, Z., Vangronsveld, K., Linton, S. J., Quartana, P. J., & Sullivan, M. J. (2012). Cognitive dimensions of anger in chronic pain. Pain, 153(3), 515-517.
17. Ferrari, R. (2015). A prospective study of perceived injustice in whiplash victims and its relationship to recovery. Clinical rheumatology, 34(5), 975-979.
18. Sullivan, M. J., Adams, H., Horan, S., Maher, D., Boland, D., & Gross, R. (2008). The role of perceived injustice in the experience of chronic pain and disability: scale development and validation. Journal of occupational rehabilitation, 18(3), 249-261.
19. Sullivan, M. J., Adams, H., Martel, M. O., Scott, W., & Wideman, T. (2011). Catastrophizing and perceived injustice: risk factors for the transition to chronicity after whiplash injury. Spine, 36, S244-S249.
20. Ferrari, R. (2014). Perceived injustice in fibromyalgia and rheumatoid arthritis. Clinical rheumatology, 33(10), 1501-1507.
21. Turk, D. C., & Okifuji, A. (1996). Perception of traumatic onset, compensation status, and physical findings: impact on pain severity, emotional distress, and disability in chronic pain patients. Journal of behavioral medicine, 19(5), 435-453.
22. Mendelson, G. (1992). Compensation and chronic pain. Pain, 48(2), 121-123
Part IIa REFERENCES:
1. Olson, S. L., & Banyard, V. (1993). " Stop the world so I can get off for a while": Sources of daily stress in the lives of low-income single mothers of young children. Family Relations, 50-56.
2. Ring, D., Kadzielski, J., Malhotra, L., Lee, S. G. P., & Jupiter, J. B. (2005). Psychological factors associated with idiopathic arm pain. J Bone Joint Surg Am, 87(2), 374-380.
3. Crichlow, R. J., Andres, P. L., Morrison, S. M., Haley, S. M., & Vrahas, M. S. (2006). Depression in orthopaedic trauma patients. J Bone Joint Surg Am,88(9), 1927-1933.
4. Systematic Literature Review of Imaging Features of Spinal Degeneration in Asymptomatic Populations. Brinjikji W, Luetmher PH et al. AJNR, 2014 Nov 27 (Epub ahead of print)
5. Lazarus, R. S., & DeLongis, A. (1983). Psychological stress and coping in aging. American psychologist, 38(3), 245.
6. Cicirelli, V. G. (1999). Personality and demographic factors in older adults' fear of death. The Gerontologist, 39(5), 569-579.
Part IIb REFERENCES:
1. Franks, P., Zwanziger, J., Mooney, C., & Sorbero, M. (1999). Variations in primary care physician referral rates. Health services research, 34(1 Pt 2), 323.
2. Mehrotra, A., Forrest, C. B., & Lin, C. Y. (2011). Dropping the baton: specialty referrals in the United States. Milbank Quarterly, 89(1), 39-68.
3. Reed, S. & Pearson, S. (2016). Retrieved 28 September 2016, from http://www.choosingwisely.org/wp-content/uploads/2015/05/ICER_Low-Back-Pain.pdf.
4. Ivar Brox J, Sørensen R, Friis A, Nygård Ø, Indahl A, Keller A, Ingebrigtsen T, Eriksen HR, Holm I, Koller AK. (2003). Randomized clinical trial of lumbar instrumented fusion and cognitive intervention and exercises in patients with chronic low back pain and disc degeneration. Spine; 28:1913.
5. Chou, R., Baisden, J., Carragee E., Resnick D., Shaffer W., Loeser J. (2009). Surgery for low back pain: a review of the evidence for an American Pain Society clinical practice guideline. Spine; 34:1094.
6. Onesti, S. (2004). Failed back syndrome. The Neurologist. 10:259.
7. Bederman S, Coyte P, Kreder J, Mahomed N, McIsaac J, Wright J. (2011). Who’s in the Driver’s Seat? The Influence of Patient and Physician Enthusiasm on Regional Variation in Degenerative Lumbar Spinal Surgery: A Population-Based Study. Spine.;36:481-9.
8. Benedetti, F. (2002). How the doctor’s words affect the patient’s brain.Evaluation & the Health Professions, 25(4), 369-386.
9. Amer Physical Therapy Assoc - Passive physical agents | Choosing Wisely. (2016).Choosingwisely.org. Retrieved 28 September 2016, from http://www.choosingwisely.org/clinician-lists/american-physical-therapy-association-passive-physical-agents-except-when-necessary/
10. Disorbio, J. M., Bruns, D., & Barolat, G. (2006). Assessment and treatment of chronic pain. Practical Pain Management, 2.
11. Schwarzer, A. C., Wang, S. C., O'Driscoll, D., Harrington, T., Bogduk, N., & Laurent, R. (1995). The ability of computed tomography to identify a painful zygapophysial joint in patients with chronic low back pain. Spine, 20(8), 907-912.
12. Jarvik, J. J., Hollingworth, W., Heagerty, P., Haynor, D. R., & Deyo, R. A. (2001). The longitudinal assessment of imaging and disability of the back (LAIDBack) study: baseline data. Spine, 26(10), 1158-1166.
13. Atlas, L. Y., & Wager, T. D. (2012). How expectations shape pain.Neuroscience letters, 520(2), 140-148.
14. Brenner, D. J. (2010). Should we be concerned about the rapid increase in CT usage?. Reviews on environmental health, 25(1), 63-68.
15. Flor, H., Fydrich, T., & Turk, D. C. (1992). Efficacy of multidisciplinary pain treatment centers: a meta-analytic review. Pain, 49(2), 221-230
16. Ritchie, C., Kenardy, J., Smeets, R., & Sterling, M. (2015). StressModEx–Physiotherapist-led Stress Inoculation Training integrated with exercise for acute whiplash injury: study protocol for a randomised controlled trial.Journal of physiotherapy, 61(3), 157.
17. Pither C., & Nicholas M. (2001). Identification of iatrogenic factors in the development of chronic pain syndromes: abnormal treatment behaviour? In Bond MR, Charlton JE & Woolf CJ (eds) Proceedings of the VIth World Congress on Pain; Pain Research and Clinical Management, vol. 4. Amsterdam: Elsevier, pp 429–434. 35.
18. Kouyanou K., Pither C., & Wessely S. (1997). Iatrogenic factors and chronic pain. Psychosomatic Medicin; 59: 597–604.
19. Alexanders, J., Anderson, A., & Henderson, S. (2015). Musculoskeletal physiotherapists’ use of psychological interventions: a systematic review of therapists’ perceptions and practice. Physiotherapy, 101(2), 95-102.
20. O'Sullivan, P. (2012). It's time for change with the management of non-specific chronic low back pain. British journal of sports medicine, 46(4), 224-227.
21. Morley, S., Eccleston, C., & Williams, A. (1999). Systematic review and meta-analysis of randomized controlled trials of cognitive behaviour therapy and behaviour therapy for chronic pain in adults, excluding headache. Pain,80(1), 1-13.
22. Burns, J. W., Kubilus, A., Bruehl, S., Harden, R. N., & Lofland, K. (2003). Do changes in cognitive factors influence outcome following multidisciplinary treatment for chronic pain? A cross-lagged panel analysis. Journal of consulting and clinical psychology, 71(1), 81.
23. Streeter, C. C., Jensen, J. E., Perlmutter, R. M., Cabral, H. J., Tian, H., Terhune, D. B., & Renshaw, P. F. (2007). Yoga Asana sessions increase brain GABA levels: a pilot study. The journal of alternative and complementary medicine, 13(4), 419-426.
24. https://www.sciencedirect.com/science/article/pii/S1053811918306256
25. https://www.sciencedaily.com/releases/2015/05/150515083223.htm
26. Retrieved from https://www.ncbi.nlm.nih.gov/pubmed/?term=Lowe a 2017 physiotherapy